Type 1 diabetes: everything you need to know!
What is type 1 diabetes?
Definition
Type 1 diabetes (T1D) was formerly called "insulin-dependent diabetes" (IDD) because it requires daily insulin injections. It is sometimes called “juvenile diabetes” because it usually appears in young patients (children, teenagers and young adults).
It is most often an autoimmune disease (the individual's immune system attacks the cells of their own body, in this particular case, the pancreatic cells that produce insulin) and a metabolic disorder at the same time (because it disrupts the transformation of proteins, lipids and especially carbohydrates, which participate in energy production).
This condition is chronic: it is characterised by a continuous excess of glucose in the blood (permanent hyperglycemia). If not controlled, it may lead to numerous complications (renal, cardiovascular, ophthalmic, etc.)
Glucose (sugar) is the fuel for the body's cells: they need it all the time to function properly (especially the brain). The glucose that circulates in the bloodstream comes from food (cereal, dairy products, fruit, etc.) or from our energy reserves (produced by the liver or the muscles).
In healthy subjects (not suffering from T1D), the pancreas secretes hormones in order to regulate the level of glucose in the bloodstream (also called glycemia). The two main hormones secreted by the pancreas are insulin and glucagon. They have opposed roles: insulin allows glucose to enter muscle cells, adipose tissue and liver, where it can be transformed (into glycogen) and stored. Thus the level of glucose decreases.
Glucagon allows the release of glucose stored in the liver, outside of meals, when blood sugar levels are falling: it increases the level of glucose in the bloodstream.
The balance of these hormones keeps blood sugar levels stabilised.
In type 1 diabetes, this regulatory system is permanently impaired. This is because pancreatic beta cells are partially or totally destroyed. There is therefore little or no secretion of insulin, which can no longer help lower blood sugar levels.
By comparison, type 2 diabetes represents the majority of diabetes cases (90%) and appears later (in people over 45). It mainly affects patients with an unbalanced diet, excessive weight or obesity. It causes progressive insulin resistance in liver cells and in the muscles that store sugar. This results in excessive insulin secretion, in the attempt to reduce hyperglycemia, which leads to the depletion of pancreatic cells and the cessation of insulin production (insulinopenia).
There is also another type 1 diabetes, called LADA (Latent Autoimmune Diabetes in Adults). It is a real T1D but its onset is late and progressive (onset at a later age than in regular T1D). It affects people under 40, without obesity or another autoimmune disease (AID). Just as the regular type 1 diabetes, it has an autoimmune component, characterised by the presence of autoantibodies in the blood (anti-GAD antibodies, in particular).
How common is type 1 diabetes?
Type 1 diabetes affects around 10% of people with diabetes in the US and around the world. Half of the cases occur before the age of 20.
Approximately 1.6 million people are currently living with type 1 diabetes in the United States, including around 200.000 children and teenagers.
For the past twenty years, the number of people with type 1 diabetes has increased by 3 to 4% per year and the onset takes place more and more early, especially in children under 5 years old. The causes of this progression remain unexplained, but environmental factors associated with genetic and nutritional factors seem to be implicated (advanced maternal age, type of breastfeeding in the first months of life, modification of the intestinal flora, exposure to toxins, etc).
Symptoms and complications of type 1 diabetes
The initial development of T1D is rapid with onset within weeks of the first symptoms:
- polyuria (abundant urine: 3 to 4 L / 24 h) and nocturia (the patient wakes up one or more times during the night to urinate);
- polydipsia (excessive thirst);
- polyphagia (excessive hunger) associated with weight loss (rapid weight loss: 9 - 22 pounds in 1 to 2 months) and muscle atrophy;
- fatigue and a lack of energy.
The symptomatology of T1D is more obvious compared to that of T2D.
T1D exposes patients to 2 major acute complications (which occur suddenly and progress rapidly):
- Hypoglycemia (abnormally low blood sugar levels) occurs in patients with T1D treated with insulin. Moderate hypoglycemia is frequent in T1D. There are various triggers: poor insulin management, reduced carbohydrate intake, unusually vigorous physical activity, kidney failure or alcohol intake. With a blood sugar level of around 3.30 mmol / L (0.60 g / L), the signs are sweaty hands, cold sweats, pale face, tachycardia, palpitations, tremors, nausea, fatigue, etc. With a blood sugar level below 2.75 mmol / L (0.50 g / L), Central Nervous System (CNS) disturbances may occur. This leads to concentration difficulties, a feeling of malaise, anxiety, confusion , drowsiness, motor coordination impairments, and can even result in coma, which can occur suddenly without any warning signs (agitated coma with convulsions). Hypoglycemia can be serious, especially if the patient is alcoholic or has kidney or liver failure.
- Diabetic ketoacidosis is a medical emergency (fatal in 5-10% of cases). The sugar no longer enters the cells due to lack of insulin. As a result, the body uses fatty acids for energy, which leads to the production of ketones (substances produced through the breakdown of fat by the liver). It develops in two phases. First, the phase of ketosis without acidosis sets in gradually: frequent urination, severe thirst, weight loss, abdominal pain, as well as nausea and vomiting. Then comes the phase of ketoacidosis which is characterised by cramps, blurred vision, severe overall dehydration, hypotension, tachycardia, acetone odour in the breath, polypnea (Kussmaul breathing), confusion, disturbances of consciousness. The patient can go into a coma, which is why it is a life-threatening emergency. This syndrome can help reveal T1D in a patient who is still undiagnosed. It can also be the result of intentional or unintentional cessation of insulin therapy (problem with the insulin pump). Finally, it can be observed in stressful situations (myocardial infarction or MI, severe infection, major surgery, etc.).
Finally, T1D is the cause of many chronic complications.
These complications are divided in two types depending on the involvement of large arterial vessels that are not specific to diabetes (macroangiopathy causing coronary heart disease, cerebrovascular disease and peripheral vascular disease), or small vessels and capillaries (microangiopathy, causing retinopathy, peripheral neuropathy and nephropathy).
Various complications can be associated with diabetes:
- cardiovascular: cholesterol deposits in the walls of blood vessels and the formation of atheromatous plaques, which lead to partial or total obstruction of blood vessels. Consequently, there is a risk of myocardial infarction (multiplied by 3 to 5 in patients with untreated type 1 diabetes), hypertension, cerebrovascular accident (stroke) and poor blood circulation in the arteries of the legs (arteritis of the lower limbs);
- kidney: diabetic nephropathy and damage to the lining of the kidneys, which allows proteins (especially albumin) to pass into the urine. Without treatment, it progresses to renal failure. Each year, 3000 diabetics start kidney dialysis or have a kidney transplant;
- neurological: hyperglycemia alters the structure of peripheral nerves (which control muscles and skin sensitivity) and nerves of the autonomic nervous system (which control various organs). This is called diabetic neuropathy;
- ophthalmic: capillary micro-occlusions (the smallest vessels) degrade the retina, which can lead to blindness;
- foot damage: insensitivity to pain exposes the diabetic to wounds in the feet which heal with difficulty and promote infections, which may result in amputations;
- sensitivity to infections: skin (abscess and gangrene), oral (gingivitis and periodontitis) and genital (urinary tract infections and vaginal yeast infection)
- liver disease: non-alcoholic steatosis or "fatty liver disease" (NAFLD / NASH);
- sexual disorders: erectile dysfunction linked to vascular involvement and diabetic neuropathy, vaginal dryness and low libido.
Diabetic patients are also more prone to infections: boils, genital candidiasis, genital pruritus ...
Causes and risk factors of type 1 diabetes
The reasons for the onset of the autoimmune reaction that causes type 1 diabetes are still unknown.
However, different factors seem to be involved:
- genetic: dysfunction of the major histocompatibility complex (or MHC, comprising the HLA genes, which help the immune system distinguish the body’s own cells from those of foreign invaders), presence of autoantigens of the β cells of the pancreas (causing their destruction );
- environmental: certain viruses (mumps, coxsackie B4, retrovirus, rubella, CMV or cytomegalovirus, Epstein-Barr), toxic substances or food (early introduction of cow's milk proteins), as well as stress, which could also cause the development of T1D;
- immunological: T1D is an autoimmune disease caused by a dysfunction in T lymphocytes (a type of cells, part of the immune system) which begin to identify the ß cells of the pancreas as foreign bodies, and to eliminate them.
Diagnosis of type 1 diabetes
The diagnosis of type 1 diabetes is confirmed by measuring blood sugar levels.
According to WHO (1999), blood sugar testing should be done after at least 8 hours of fasting.
Normal fasting blood sugar values are 0.7-1 g / L (3.9-5.5 mmol / L).
In a healthy person, it should be less than 1.1 g / L (6.1 mmol / L).
Diabetes corresponds to chronic hyperglycemia defined by:
- fasting blood glucose level ≥ 1.26 g / L (7 mmol / L), measured twice;
- blood sugar level ≥ 2 g / L (11.1 mmol / L) at any time of the day;
- blood glucose level 2 hours after taking 75 g of oral glucose (oral hyperglycemia) ≥ 2 g / L (11.1 mmol / L).
The latter is not recommended for diagnosis and should become exceptional because there is a risk of developing retinopathy within 10 to 15 years.
For the diagnosis of typical cases, no need for additional laboratory tests (HLA, auto-Ac etc).
On the other hand, for slow T1D (LADA: Latent Autoimmune Diabetes in Adults), detection of the presence of anti-GAD antibodies is necessary.
After the diagnosis of T1D, treatment must begin without delay.
Treatments for type 1 diabetes
Insulin is the essential replacement treatment for patients with type 1 diabetes, as there is no pancreatic insulin secretion. It is called insulin therapy.
The aim of the treatment is to control blood sugar levels, prevent hyperglycemia and hypoglycemia, as well as vascular complications, by limiting the irreversible binding of glucose to proteins (membrane glycation).
Daily insulin requirements (total daily insulin dose, or TDI) are between 0.7 and 0.8 U / kg , but they vary depending on the patient and their physical activity.
Half of the insulin dose is necessary for basic insulin production (to ensure proper functioning of the body) and the other half is taken with food or drink.
Different types of insulin
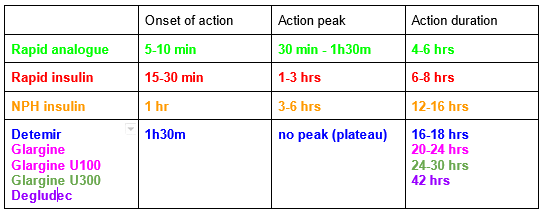
Three important pharmacological parameters make it easy to differentiate insulins: onset of action, period of maximum activity and end of action.
The different types of insulin usually have a concentration of 100 IU / mL (but some are concentrated at 200 IU / mL or 300 IU / mL). Among them, there are:
- short-acting insulin, which corresponds to rapid human insulin or to rapid insulin analogues;
- basal insulin which can be intermediate (Neutral Protamine Hagedorn or NPH with intermediate duration), slow, like insulin glargine (Lantus®) or insulin detemir (Levemir®), or ultra-slow like insulin degludec (Tresiba®) ) and insulin Glargine U300 (Toujeo ®);
- mixtures which can be traditional (rapid / NPH), or combine rapid analogues / NPH.
Rapid human insulin (rapid Humulin® and Actrapid®) is a colourless solution. The time between the injection and the meal is approximately 30 minutes. It comes in the form of pens or vials (solution).
The disadvantages of rapid human insulin is that it has a very variable duration of disappearance from the blood (duration of absorption) depending on the individual.
This is why rapid or even ultra-rapid insulin analogues have been developed, their subcutaneous absorption is accelerated (their effect is immediate).
These include insulin lispro (Humalog®), insulin aspart (Novorapid® or Fiasp®) and insulin glulisine (Apidra®), all are colourless solutions. They do not require a delay between the injection and the meal (faster and shorter blood sugar lowering effect). They also come in pens or bottles.
NPH insulin (Neutral Protamine Hagedorn) is a protein (protamine) that binds human insulin, then releases it gradually (Humulin® NPH and Insulatard®). It is a suspension (dispersion of solid in a liquid, with a milky colour). It comes in the form of a mixture, but also in the form of pens or bottles.
Slow insulins, such as detemir (Levemir®), glargine (Lantus®, Abasaglar® or Toujeo®) and degludec (Tresiba®), are intended to reduce the variability of blood sugar levels and prevent nocturnal hypoglycemia.
The characteristics of these different types of insulins are summarised in the following table:
The rapid human insulin + NPH insulin mixtures (Humulin profile® 30), composed of 30% rapid human insulin and 70% NPH insulin, can be in the form of pens or vials.
But these mixtures are gradually abandoned in favour of mixtures with rapid analogues (analog of insulin + protamine which slows down the action). They allow better glycemic stability and improve patients’ quality of life:
- Humalog Mix® 25, 50
- Novomix® 30, 50, 70 (with 25/30/50/70 which correspond to % of insulin analog).
Intensive insulin therapy
In terms of treatment for T1D, it is always necessary to combine a basal insulin and a rapid insulin. The basal-bolus regimen with 4 injections (at breakfast, lunch, dinner and before bed) is the most common. It combines the use of:
- rapid analog bolus (breakfast, lunch and dinner) aimed at achieving the level of blood glucose at <1.6 g / L 1 hour after a meal;
- and basal insulin (slow, such as Lantus® which requires 1 injection, Levemir® which requires 1 to 2 injections, or intermediate insulin which requires 2 injections, or a fast analogue in a pump) in the evening before going to bed, the adjustment of the dose is made according to the fasting glucose level in the morning, with a target of 0.7 to 1.1 g / L.
The patient adapts their insulin treatment to their lifestyle (diet, physical activity) rather than the other way around. To succeed, it is important that the patient is able to assess his or her carbohydrate intake and level of physical activity.
>> To learn more about intensive insulin therapy, click here to read our article <<
Insulin storage
It is important to mention that :
- insulin is destroyed at temperatures below 32° F;
- insulin gradually loses its effectiveness at temperatures above 104° F.
In addition, it is recommended to store insulin in the crisper drawer of the refrigerator, and not in the door, to avoid loss of effectiveness by inadvertent shaking.
Before the 1st use, cartridges, vials and pens can be stored until the expiration date at a temperature between +35.6 and + 46.4° F.
Once opened, cartridges and pens can be stored for 4 weeks, compared to 6 weeks for bottles. They should be stored at room temperature (<77° F) and protected from sunlight. In fact, it is not recommended to keep the insulin in a cool place once it has been opened, in order to avoid thermal shocks which may alter the product.
Insulin devices
There are different types of insulin devices:
- single-use insulin syringes with graduation in international units (IU);
- reusable or disposable injection pens (Solostar, Flexpen, Innolet, Kwikpen);
- refillable pens which are useful in pediatrics (with ½ unit graduations);
- insulin pumps.
Injecting insulin is easier with a pen than with a syringe. Insulin is available at all times in a refillable pen fitted with a cartridge or in a disposable pre-filled pen. They allow you to select insulin by ½, 1 or 2 unit (s).
Needle sizes vary depending on the situation: 4mm needles are recommended in pediatrics.
In order to place the needle on the pen, you must:
- Use a new needle for each injection, check that the cap is intact and remove it;
- Place the needle in the same axis as the pen, pierce the membrane of the pen with the internal needle, screw the needle onto the pen (try not to tighten too much);
- Remove the outer and inner caps.
As for insulin syringes, different types are used depending on the injection dose:
- 0.3 ml syringes containing 30 units of insulin
- 0.5 ml syringes containing 50 units of insulin
- 1 ml syringes containing 100 units of insulin
The syringe is filled with either a single insulin or a mixture of short-acting and long-acting (NPH) insulin.
Finally, insulin pumps improve blood sugar levels and the patient's quality of life (no need for frequent pricks). There is a fixed or variable basal rate, with a bolus at mealtime. These are either external pumps with a subcutaneous catheter or pumps implanted with an intraperitoneal catheter (in the abdominal cavity) which allows regular and repeated blood glucose self-monitoring. But it is still necessary to have disposable insulin pens in case of failure of these devices.
The catheter should be adapted according to the patient’s age and thickness of the subcutaneous tissue:
- shorter perpendicular catheters should be used by adults, in order to avoid intramuscular injection (more painful), but are unsuitable for small children and children with little subcutaneous tissue;
- tangential catheters are recommended for all children under 6 years old, for thin or athletic children (because there is a greater risk of the catheter being removed) and in the event of a complication at the infusion site (infection, redness, recurrent obstructions).
Administration rules
For insulin in suspension (NPH): agitate slowly, with vertical movements, 10 times. You should then check that the insulin colour is white / opaque.
For all types of insulin, you must:
- Purge the pens before each injection (1-2 units) by directing the needle upwards
- Inject into clean skin, without prior disinfection with alcohol because it causes intolerance at the injection sites
- Do not inject through clothes.
- The speed of insulin release varies depending on the injection site (abdomen> arms> thighs and buttocks). Do not withdraw the needle too quickly because there is a risk of insulin loss (hold the needle for 10 seconds).
Rotation of the injection sites is necessary to avoid the risk of lipodystrophy (excess of lipids). The latter can cause pain at the injection site and decreased insulin absorption (and therefore metabolic control).
Living with type 1 diabetes
Type 1 diabetes requires the use of suitable equipment (for injections and for self-monitoring). Therapeutic patient education (by the attending physician, diabetologist or pharmacist) is essential for optimal blood sugar control. The patient and their family must be well-informed and know how to deal with possible hypoglycemia and acute complications.
Self-monitoring of blood glucose levels
Self-monitoring of blood glucose is fundamental, treatment should be adapted to the patient’s diet and physical activity.
It is performed before a meal (pre-meal), then 1 to 2 hours after a meal (post-meal), as well as at bedtime.
Using finger glucose monitors (such as Accu-Chek® and OneTouch®) requires:
- washing your hands in hot water, drying them carefully and not using alcohol
- massaging your fingers to activate blood circulation
- pricking fingers on the sides (avoid index and thumb), and for a less painful dextro, it is advisable to change the site of pricking every time you prick.
- disposing of the used equipment in a suitable container. All prickly, sharp materials used in the treatment of diabetes (including lancets, lancing devices, syringes, pens with needles, catheters and needles alone) must be sorted by patients and stored in needle boxes.
>> Click here to read our article about disposing of used medications
Keeping a journal (or using a mobile application) can be helpful for blood glucose self-monitoring.
Today, continuous glucose monitoring (CGM) is possible and represents a revolution in the management of diabetes. A subcutaneous sensor (such as FreeStyle® Libre or MiniMed®) is installed, making it possible to measure glucose level in the interstitial fluid (the space between the blood capillaries and the cells).
Urinary self-monitoring (glucose, ketone bodies, proteins) is sometimes necessary for children, pregnant women, pump carriers or patients with unexplained hyperglycemia.
The measurement of glycosuria (glucose level in the urine) should be performed if the capillary blood sugar level is > 1.8 g / L:
- if the values of ketonuria (levels of ketone bodies in the urine) and glycosuria are zero (negative) then blood sugar levels are <1.8 g / L during the preceding period;
- if ketonuria is negative and glycosuria is positive, then the blood sugar levels have exceeded the renal threshold (generally 1.8 g / L) but the imbalance is not major;
- if ketonuria and glycosuria are positive, then the patient has had more severe hypoglycemia and insufficient insulin for part of the night;
- if ketonuria is positive and glycosuria is negative, then the blood sugar levels remained normal during the night but the food intake from the night before was insufficient.
Monitoring long-term glycemic balance
Fasting blood sugar testing should be done once or twice a year at a hospital.
Furthermore, in diabetes, glycation (irreversible binding of glucose to proteins) increases with blood sugar. The concentration of glycated proteins thus reflects variations in blood sugar levels over several months (this is referred to as “glycemic memory”).
Measuring the level of glycated hemoglobin (HbA1c) is an essential test for patients with diabetes.
HbA1c values reflect the average blood glucose values over the past 2 months. It is normally <6% of total hemoglobin (Hb).
In adults with diabetes, it should be ≤ 7%. In children and the elderly (> 75 years old), the glycemic target may be higher, given the risk of severe hypoglycemia (it is established by the doctor and adapted to each patient).
The frequency of the HbA1c testing is:
- every 6 months if the goal is reached and the treatment is not changed.
- every 3 months in other cases (for example if the balance is insufficient or during change of treatment).
The HbA1c value is independent of weight and exercise and increases moderately with age.
There are, however, some precautions to be taken, particularly in the event of hemolysis, hemoglobinopathy, transfusions or chronic renal failure.
The HbA1c test is useful for monitoring diabetes, but not for diagnosing it.
Detecting fructosamines can also be useful when the values of HbA1c are difficult to interpret (haemolysis, haemoglobinopathies, etc.) or in the event of a need for closer monitoring (pregnancy, change of treatment, etc.). They can be detected by measuring the total glycated serum proteins. This test reflects the blood sugar balance over the previous 20 days.
Screening for complications of type 1 diabetes
Monitoring the risk factors of cardiovascular disease is necessary in patients with diabetes. Atherosclerosis (formation of atheroma plaques and obstruction of large and medium arteries), for example, is very common in diabetics.
An EAL (Exploration of a Lipid Anomaly) must be carried out once a year with the determination of total cholesterol (CT), triglycerides (TG), cholesterol linked to HDL (good cholesterol) and cholesterol linked to LDL (bad cholesterol).
Qualitative abnormalities of lipoproteins (lipid transport proteins in the blood) can be observed: VLDL (Very Low Density Lipoprotein), which is larger and richer in triglycerides (TG) and an increase in small and dense LDL (Low Density Lipoprotein, which causes the formation of atherosclerotic plaques) are important atherogenic factors.
Screening for and monitoring of renal complications (nephropathy followed by chronic renal failure) is also necessary.
Therefore, once a year, it is important to measure the levels of:
- serum creatinine (blood creatine level) with estimated creatinine clearance (according to the CKD-Epi or MDRD formulas).
- microalbuminuria (low but abnormal amount of albumin in the urine).
The normal values of albuminuria are <30 mg / 24h (2 mg / mmol) and a microalbuminuria between 30 and 300 mg / 24h (2-20 mg / mmol of creatinine) should lead to increased control of blood sugar and blood pressure. The determination of the level of microalbumin in the urine is an essential examination because it is the first abnormality that indicates kidney damage.
Finally, the search for hematuria (presence of blood in the urine) and CBEU (cytobacteriological examination of the urine which makes it possible to identify the germ causing a urinary infection) must also be carried out annually.
>> For more information, read our article “Diabetes: how to read blood tests”
Screening for other complications can also be useful:
- annual cardiovascular examination (ECG at rest, monitoring dorsalis pedis (foot) and posterior tibialis (ankle) pulse, searching for carotid, femoral or abdominal bruits (murmurs), etc.).
- annual ophthalmologic check-up to detect retinopathy;
- examining for signs of peripheral neuropathy (neurological complications) once a year;
- annual examination of the foot (small lesions, trophic disorders, cracks, etc.).
Dietary and lifestyle recommendations
It is important to split your carbohydrate intake during the day (20% at breakfast and 40% at lunch and dinner, or 30%, if there is a snack - 10%) and to opt for foods with a low glycemic index <50, such as apples, pears, oatmeal, cereal bread, peas, lentils, dark chocolate or skimmed milk. The glycemic index is the rate at which food can increase a person's blood sugar level.
Carbohydrate intake should also be synchronised with insulin therapy. This means that you have to inject yourself with insulin every time you eat food that contains sugar (carbohydrates).
Portion control is necessary: carbohydrates should make up 50-55% of total daily energy intake, fat - 35-40%, and protein 11-15%. The intake of 25 to 30 g of fiber per day (as in apples or cereal, for example) is also advisable.
It is recommended to vary the sources of protein: animal proteins (rich in amino acids and iron) and vegetable proteins (rich in starch and micronutrients). It is also necessary to limit fatty meats, consume 3 dairy products per day, as well as legumes (lentils, white beans, flageolet beans ...) and cereal.
Saturated fatty acids should be limited (butter, cream, cold cuts, etc.), and monounsaturated (omega 9 contained in olive oil) and polyunsaturated fatty acids (omega 3 contained in fatty fish including mackerel, sardines and cod liver) should be part of your daily diet.
Moreover:
- foods and drinks high in added sugar (sodas) should be avoided (<10% of total daily energy intake);
- alcohol should be consumed moderately if diabetes is well managed (as well as carbohydrate intake), otherwise it should also be avoided.
What to eat when you have hypoglycemia?
Managing hypoglycemia is essential. When symptoms appear (paleness, sweating, hunger, fatigue, blurred vision, dizziness, headaches, change of character, etc.), you must act quickly.
If the patient is conscious, they should take either 3 lumps of sugar, or 1 small box of fruit juice, or 1 tablespoon of honey or jam or 1 fruit jelly candy. It is also recommended to always have 1 lump of sugar on you to prevent hypoglycemia.
Blood sugar level should be checked 15 minutes after:
- If it is still <0.7 g / L (or 4 mmol / L) and / or the symptoms are present: repeat the actions;
- If it goes back to normal (between 0.7 and 1.1 g / L) but the next meal is in more than 2 hours: it is possible to have a snack (fruit, milk, yogurt or bread );
- If it is back to normal and a meal is planned soon, then no snack should be taken.
Finally, if the patient is unconscious, glucagon 1 mg (Glucagen®) must be injected intramuscularly (IM), and if this fails, repeat the injection after 15 minutes.
It is also important to understand the origin of hypoglycemia, that is, to ask yourself:
- if the dose of insulin used was too high or if the treatment is inappropriate;
- if food intake has been insufficient or delayed;
- if you have had unforeseen physical activity, or it was too vigorous.
It may be useful to keep a hypoglycemia journal and you should not hesitate to talk to your doctor about it.
Physical activity
Regular physical activity is an essential part of diabetes treatment. By physical activity we mean any type of activity that causes muscles to contract and thus increases energy expenditure. It includes daily household activities and chores (cleaning, walking the dog, gardening, mowing the lawn, playing in the park with the children, etc.). In addition, certain changes in habits, when possible, allow you to have more exercise on a daily basis, such as taking the stairs instead of the elevator or escalator, or getting around on foot or by bike instead of taking public transport or the car.
Physical activity improves muscle capacity and exercise endurance, helps reduce the risk of cardiovascular disease, and control cholesterol and weight, promotes good-quality sleep and reduces stress. Finally, it increases the consumption of glucose in the blood and thus allows better control of blood sugar levels and of diabetes in general.
Recommendations in terms of physical activity are the same for the general population and for diabetics: the WHO recommends 150 minutes of endurance sport of moderate intensity per week (such as, for example, brisk walking, cycling , swimming…). The best thing is to practice an average of 30 minutes of endurance sport every day.
It is advisable to make an appointment with your GP before starting or resuming physical activity in order to choose a suitable activity and to take the necessary precautions, allowing you to perform it safely. It is best to start with low-intensity activities (slow walking, cleaning, gardening, etc.), and then gradually increase, if possible, their intensity and duration.
Pregnancy
If a woman has diabetes, it is better to get pregnant earlier in life, before any complications arise. It is therefore important to carefully plan your pregnancy.
It is recommended that you achieve the best possible blood sugar control before and during pregnancy, with the lowest possible HbA1c. It is advisable to set up a follow-up care plan together with diabetes specialists and obstetricians.
Inheritance is low for type 1 diabetes with a risk of 2-3%, if the mother has diabetes.
However, with T1D there is an increased risk of acute complications (hypoglycemia, ketoacidosis) and a risk of worsening of such conditions as diabetic retinopathy and nephropathy.
There is an increase in frequency of:
- high blood pressure during pregnancy, called gestational hypertension;
- pre-eclampsia (high blood pressure and the presence of protein in the urine, which can cause serious complications);
- spontaneous miscarriages;
- prematurity;
- cesarean section.
Maternal diabetes can also cause foetal malformations (especially during the first trimester of pregnancy). However, if diabetes is recent and well managed, the risk of foetal malformations is close to that in “normal” pregnancies.
An increased risk of foetal and neonatal complications may also be observed:
- macrosomia (birth weight greater than 8.8 pounds);
- an enlarged heart septum (excessive thickening of the septum (wall) that separates the chambers of the heart);
- tissue hypoxia (insufficient supply of oxygen to the tissues). It is associated with
- polycythaemia (increase in haematocrit, or the ratio of the volume of red blood cells to the total volume of blood) and hyperbilirubinemia (increase in the level of bilirubin in the blood, a yellow pigment resulting from the breakdown of haemoglobin, the protein of red blood cells that carries oxygen) in newborns;
- delayed pulmonary maturity (possible neonatal respiratory distress);
- hypoglycemia of the newborn, should be carefully monitored.
Thus, glycemic control is stricter in case of diabetic pregnancy and requires supervision (attending physician, diabetologist, etc.). Treatment can be adapted or modified to keep blood sugar levels under control, because insulin requirements increase significantly, especially around the 6th month of pregnancy, and almost double at the end of pregnancy. Using an insulin pump can be very effective in controlling blood sugar fluctuations.
To conclude, type 1 diabetes is an autoimmune disease characterised by lack of insulin production, which causes an excess of glucose in the blood. Scientists are still searching for possible reasons for this dysfunction.
Treatment for type 1 diabetes consists of insulin injections.
New technologies, such as continuous blood glucose monitoring systems, are making it possible to improve patients’ quality of life. Finally, innovative treatments can be used, such as artificial beta cells or closed-loop control systems, or transplants of the Islets of Langerhans (pancreatic cells) in patients with poorly controlled type 1 diabetes.
Sources :
https://www.ajd-diabete.fr/le-diabete/tout-savoir-sur-le-diabete/le-traitement/
https://infos-diabete.com/diabete/diabete-de-type-1/
https://www.inserm.fr/information-en-sante/dossiers-information/diabete-type-1
https://www.federationdesdiabetiques.org/information/diabete
https://www.federationdesdiabetiques.org/information/diabete/chiffres-france
https://www.ameli.fr/assure/sante/themes/diabete-comprendre/definition
https://www.ameli.fr/assure/sante/themes/activite-physique/diabete-activite-physique-fondamentaux
https://www.has-sante.fr/upload/docs/application/pdf/ald8_guidemedecin_diabetetype1_revunp_vucd.pdf
https://www.has-sante.fr/upload/docs/application/pdf/bd-07-057-diabete_1_mar_08.pdf
http://www.sfendocrino.org/article/392/item-233-a-ndash-diabete-sucre-de-type-1
https://www.who.int/fr/news-room/fact-sheets/detail/diabetes
https://www.chuv.ch/fileadmin/sites/dedop/documents/vivre-avec-diabete.pdf
https://www.ajd-diabete.fr/le-diabete/tout-savoir-sur-le-diabete/ladministration-dinsuline/
https://www.ajd-diabete.fr/le-diabete/tout-savoir-sur-le-diabete/la-pompe-a-insuline/
Published Jul 19, 2019 • Updated Dec 28, 2021



