Breast cancer: definition, symptoms, treatment
What is breast cancer?
Definition
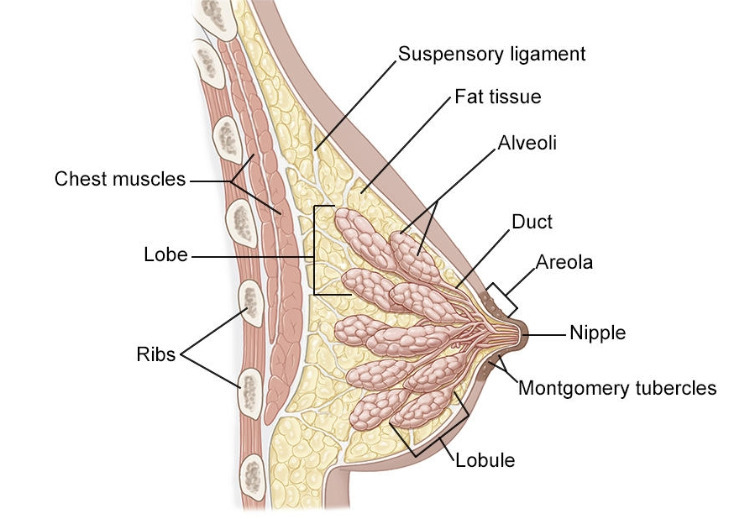
The breast (also called the mammary gland) is made up of different compartments, which consist of:
- lobules, where the synthesis of milk takes place;
- and milk ducts, which collect milk and lead it to the nipple.
These compartments are surrounded by a supportive tissue, made up of fat, fibers, and blood and lymphatic vessels.
Source: Breastfeeding counselor networking program
Breast cancer is the most common and deadly cancer affecting women in the USA. It results from malfunction of the cells, which multiply and usually form a mass called a tumor.
There are different stages of the disease:
- among benign tumors (called adenofibromas), are identified:
- adenofibroma or fibroadenoma: it is a tumor made up of connective and glandular tissues, it can be detected via mammography and ultrasound. It never causes axillary lymphadenopathy and requires monitoring or surgery if the mass is greater than 30 mm.
- fibrocystic breast disease: it is caused by the development of one or more calcified cysts, and requires surgery.
- among localized cancers (at the level of the ducts and lobules, without lymph node involvement), corresponding to stages 0 to II, we can distinguish:
- carcinoma in situ: ductal or lobular (respectively 4% and 2.5% of breast cancers), without crossing the basement membrane. Detected by mammography and confirmed by biopsy, there is a 33% risk of developing invasive carcinoma within the next 10 years.
- invasive carcinoma: ductal or lobular (respectively 75% and 13% of breast cancers), with invasive involvement of neighbouring tissues. Two thirds of cancers are detected by mammography at this stage.
- Cancer is locally advanced (stage III) when cancer cells have reached either the axillary nodes or the internal mammary nodes located in the chest wall.
- Cancer is metastatic (stage IV) when cancer cells have invaded the breast and entered neighbouring lymphatic or vascular vessels. The main metastatic locations are: osseous, or bone (50%), locoregional (40%), hepatic (30%), pulmonary or pleural (20%) and cerebral (10%).
Breast cancers are mostly hormone-dependent, depending on estrogen (ER +). Some are dependent on the overexpression of HER2 (Human Epidermal growth factor Receptor 2), which is a transmembrane receptor involved in the regulation of cell proliferation. The others are said to be triple negative because ER-, HER2- and do not have a characteristic surface marker.
Is breast cancer common?
It is the most common cancer in women with 50,000 new cases per year. It affects 1 in 10 women and has a female to male sex ratio of 99 (breast cancer exists in men). It is a rare cancer in women under 30, with a peak in cancer development between the ages of 60 and 69. Relative 5-year survival rate is 88% and 10-year survival rate is observed in 80% of localized forms. Nevertheless, it is the leading cause of death from cancer in women, with a mortality rate of over 40 thousand deaths per year in the US.
Symptoms and complications of breast cancer
Symptoms of breast cancer are:
- the appearance of a lump in the breast, fixed or mobile, often with irregular contours, seen during self-examination;
- swollen and hard lymph nodes in the armpit, not painful;
- a change in the shape of the breast;
- a change in the appearance of the skin of the breast (redness, edema or orange peel skin) or of the nipple or areola which is the area surrounding the nipple (retracts, changes colour, etc.);
- more rarely, spontaneous discharge from the nipple;
When breast cancer has spread to the rest of the body, other symptoms may appear, such as fatigue, nausea, weight loss, bone pain, and blurred vision.
Causes and risk factors of breast cancer
Among the risk factors for breast cancer, there are personal factors, such as:
- hormonal factors, which correspond to hyperestrogenism:
- Precocious puberty before the age of 12;
- Late menopause after the age of 50;
- Being a nulliparous woman (who has never given birth): the risk of developing breast cancer decreases with the number of pregnancies and increases with the late nature of the first pregnancy (> 35 years);
- Hormone replacement therapy for menopause that lasted more than 10 years;
- Postmenopausal obesity (estrogen is also produced by fat cells).
- age: there is a peak in the risk of developing breast cancer, between the ages of 60 and 64.
There are also family or genetic risk factors:
- when several women from the same family (first and second degree) have (or have had) breast cancer;
- when a woman in the same family had breast cancer before the age of 40;
- when several people in the same family have (or have had) colon or ovarian cancer.
Among the genetic predispositions, we can cite:
- mutation of the BRCA1 and BRCA2 predisposing genes, present in 5 to 7% of breast and / or ovarian cancers
- mutations in other genes (p53, PI3K, PALB2).
Finally, there are behavioural risk factors, which can be:
- food-related (excessive consumption of animal fats, sugars and alcohol);
- or related to smoking;
Diagnosing breast cancer
Screening
Self-examination is a simple procedure to be performed once a month by all women, always at the same time, that is, a few days after the end of period. It allows you to detect an anomaly rather early and report it to your doctor. The self-examination consists of standing or sitting in front of your mirror and inspecting the appearance of your breasts (fluid discharge from the nipple, dimples, orange peel skin ...). Then you should raise an arm and carefully and firmly palpate the breast with the three fingers of the opposite hand, making small circles, from the outer part of the breast (paying particular attention to the area between the armpit and the breast) to the nipple (and thus verify the absence of lump or thickening).
Mammography is a standard radiographic examination, bilateral and comparative. The signs of breast cancer that can be observed are opacity, architectural disorganization or microcalcifications.
There is a free breast cancer screening programme for women aged 50 to 74, which consists of a mammogram every 2 years.
Ultrasound is a second-line examination, complementary to mammography, performed when the breasts are dense, or to assess the abnormality detected on mammography.
Diagnosis
The diagnosis of breast cancer consists of several steps:
The diagnosis of breast cancer consists of several steps:
- first of all, there is an interview with the patient, in order to determine possible risk factors;
- a clinical examination (palpation of the nodules) is then carried out, in search of axillary lymphadenopathy;
- then comes medical imaging: mammography, ultrasound of both breasts and lymph nodes, MRI, PET (positron emission tomography) scan;
- finally, mammography or ultrasound-guided biopsies are carried out in order to establish a histological diagnosis which alone makes it possible to assess the elements of prognosis and the treatments to be considered.
In the event of invasive localized cancer, an extended assessment may be performed: bilateral mammogram, liver ultrasound, abdomen and pelvis scan, chest X-ray, bone scintigraphy (bone scan), MRI and brain scan (if neurological symptoms are present), or even a FDG PET scan.
TNM classification
This classification is used to choose the best therapeutic strategy.
- T = Size:
- T1: if the size of the tumor is <2 cm;
- T2: if the size of the tumor is between 2 and 5 cm in its largest measurement (starting from T2, a histology (tissue analysis) is essential to decide on the treatment);
- T3: if the size of the tumor is > 5 cm in its largest measurement;
- T4: if the tumor, whatever the size, presents direct extension to the chest wall (T4a) or to the skin (T4b);
- T4d: if it is an inflammatory carcinoma.
- N = Regional lymphadenopathy (N for “nodes”):
- Nx: if there is no information on lymphadenopathy;
- N0: if there is no sign of regional lymph node invasion;
- N1: if axillary mobile ipsilateral lymphadenopathy is observed (invasion of 1 to 3 nodes);
- N2: if axillary lymphadenopathy (N2a) (4 to 9 lymph nodes) or ipsilateral mammary lymphadenopathy (N2b) are observed, fixed together or hardened;
- N3a: if ipsilateral subclavian lymphadenopathy is observed with axillary lymphadenopathy (> 10 lymph nodes);
- N3b: if there is an ipsilateral internal mammary lymphadenopathy with axillary lymphadenopathy;
- N3c: if ipsilateral supraclavicular lyphodenopathy is observed
- M = Metastases:
- M0: if there are no metastases;
- M1: if metastases are present.
Pathological examination
Scarff, Bloom and Richardson histoprognostic grade: SBR
The SBR score is established based of 3 criteria:
- the degree of glandular/tubular differentiation of the tumor;
- irregularity of the size of the nuclei;
- the mitotic index (number of mitoses).
Are then evaluated:
- the histological appearance: well (1), moderately (2) or poorly differentiated (3);
- the appearance of the nucleus: weak (1), medium (2) or strong (3) anisonucleosis (all the nuclei of the cells in a tissue are not the same size);
- the number of mitoses over 10 fields: absence (1), a little <12 (2) or a lot> 12 (3).
Thus, the total of the scores gives the SBR grade:
- SBR I: scores 3, 4 or 5 for less aggressive tumors;
- SBR II: scores 6 or 7 for moderately aggressive tumors;
- SBR III: scores 8 or 9 for aggressive tumors.
Tumor markers
There are different tumor markers for breast cancer:
- hormone receptors (RH or ER) for estrogen RO or progesterone RP can be detected by immunohistochemistry. This makes it possible to know whether there is, or isn’t, a hormone dependence, knowing that ER + breast cancers are less aggressive;
- the HER2 oncogene (Erb-B2) encodes EGFR receptor 2 (HER2), overexpression of which stimulates cell growth. Overexpression of this oncogene is thus observed in 30% of metastatic breast cancers, given that HER2 3+ breast cancers are highly aggressive;
- the percentage of cells in S phase (DNA replication phase) can be calculated by flow cytometry;
- finally, the Ki index 67 is an evolutionary score of prognostic interest (progression-free survival and overall survival), Ki being a cell proliferation gene.
Treatments for breast cancer
Surgery
Surgery does not only remove the tumor, but also confirms the diagnosis and specifies the stage of cancer development by assessing the invasion of the axillary lymph nodes.
There are two types of surgery for breast cancer:
- lumpectomy, that is, conservative surgery, performed if the tumor is well localized and is less than 3 cm (without aesthetic consequences);
- total mastectomy (1/3 of all cases) if the tumor is large and multifocal or if a relapse is observed after conservative treatment.
In uninfected women carrying a BRCA1 / 2 mutation, prophylactic bilateral mastectomy is, despite its mutilating nature, the most effective cancer risk prevention measure.
In addition, the search for the "sentinel lymph node" allows, in case of small tumors, to remove only certain nodes (the first lymph nodes that drain the tumor). They are identified by the injection of a dye solution (patent blue) in the vicinity of the tumor. The lymph node is analyzed intraoperatively:
- if negative (not affected by cancer cells): axillary dissection is not performed, and there are therefore fewer complications;
- if positive: 8 to 10 nodes are removed.
Finally, immediate reconstruction after mastectomy is not recommended if radiotherapy is planned.
Radiotherapy
Carried out 4 weeks after the surgery, radiotherapy is called "adjuvant". Its aim is to minimize the risk of a local relapse, and it increases the overall survival rate. 4 areas can be treated by radiotherapy:
- the mammary gland after lumpectomy;
- the tumor bed (that is, the area of the breast where the tumor was located before surgery);
- the chest wall after mastectomy;
- lymph node areas close to the tumor.
The cumulative radiation dose is 50 Gy, or 2 Gy a day, 5 days a week for 5 weeks.
Radiation therapy is not used in case of a relapse if it has already been done the first time.
Possible complications are rash, swelling of the breast, pain, fatigue, and long-term cardiovascular and lung problems.
Systemic medical treatments
They can be of 3 types:
- hormone therapy corresponds to treating RH + (ER +) cancers by inhibiting hormonal stimulation;
- chemotherapy is the inhibition of tumor growth by conventional cytotoxic chemotherapy;
- targeted therapies correspond to the inhibition of tumor growth by inhibiting neovascularization, restoring apoptosis (programmed cell death) or stimulating the patient's immune response.
Hormone therapy
Its aim is to increase relapse-free survival and overall survival after surgery and / or radiotherapy.
In pre-menopausal women, inhibition of LH production can be achieved with LH-RH analogues, such as goserelin (Zoladex®) or leuprorelin (Prostap®, Lutrate), which are implants injected subcutaneously (SC) for prolonged administration (1 month). They are indicated in cases of RH + (ER +) and HER2 +++ breast cancer, or if there is a gynecological indication with endometriosis and uterine fibroma. The side effects that may be observed are headaches, mood disturbances, hot flashes, sweating attacks, decreased libido, joint pain, skin reactions, high blood pressure or the risk of fractures.
Alternatively, competitive estradiol inhibitors can be used, such as tamoxifen 20 mg / day. The latter is indicated in RH + (ER +) and HER2- breast cancers in premenopausal women. 5 years of treatment reduces the risk of a relapse by 45 to 8% and the risk of death by 32 to 10%. Immediate intolerance can be observed and consists of hot flashes, vaginal pruritus (itching), leucorrhea (bloodless vaginal discharge) and nausea. It also increases the risk of thromboembolic events of the order of 1% (stroke, embolism), endometrial cancer by 0.4 to 1.7% (requiring gynecological monitoring) and cataracts. There are also drug interactions with CYP 2D6 inhibitors (paroxetine, fluoxetine, quinidine should therefore be avoided if treatment with tamoxifen).
In addition, fulvestrant (Faslodex®) can also be used as a second line treatment in postmenopausal women.
In postmenopausal women, aromatase inhibition is achieved. The latter is present in tumor tissue, adrenals, muscles, adipose tissue, liver (and in the ovaries and breasts in postmenopausal women). Its inhibition leads to a decrease in the synthesis of circulating estrogens and estrogen receptors (ERs). These aromatase inhibitors can be:
- competitive (steroids): formestane, exemestane (Aromasin®);
- non-competitive (non-steroidal): letrozole (Femara®), anastrozole (Arimidex®).
They are all administered orally and indicated for ER + breast cancer in postmenopausal women. Slightly to moderately severe side effects that may be observed are hot flashes, hair loss, digestive disorders, asthenia, drowsiness, headaches or rash. Bone and joint pain may also be observed and the risk of osteoporosis is increased compared to tamoxifen.
Chemotherapy
Its aim is to prevent tumor growth by interfering with the synthesis of nucleic acids (DNA and RNA), blocking cell division, inhibiting neovascularization of tumors, inducing or restoring apoptosis (self-destruction of cells) and stimulating the immune response.
The molecules used all have an influence on DNA and can be:
- antimetabolites: 5-fluorouracil (5-FU) or capecitabine;
- alkylating agents: cyclophosphamide (CPM);
- intercalating agents or topoisomerases type 1 and 2 inhibitors: anthracyclines (epirubicin, doxorubicin).
These 3 molecules are combined in the FEC protocol (fluorouracil, epirubicin and cyclophosphamide).
- spindle poisons or antimitotics: among which can be found polymerization inhibitors (vinca-alkaloids, such as vinblastine, vincristine or vinorelbine) and depolymerization inhibitors (taxanes, such as paclitaxel and docetaxel).
The common side effects to all these treatments are:
- haematological (neutropenia, thrombocytopenia, anaemia);
- digestive (nausea and vomiting);
- stomatological (stomatitis, mucositis, or inflammation of the mucous membranes);
- dermatological (alopecia, or hair loss)
- gynecological (amenorrhea, or absence of periods).
Some side effects are drug-specific:
- oral and throat mucositis (inflammation, burns, canker sores and yeast infection) and hand-foot syndrome for 5-FU or capecitabine;
- urotoxicity (haemorrhegic cystitis) for cyclophosphamides;
- dose-dependent cardiotoxicity for epirubicin / doxorubicin;
- skin / inguinal edema, skin erythema (prevention with an oral steroid the day before and for 2 days) for docetaxel;
- neuropathy and myalgia (prevention with an oral steroid) for paclitaxel;
- neuropathies (paresthesias) for vinorelbine.
Targeted molecular therapy
Different molecules can be used:
- Bevacizumab (Avastin®) is a monoclonal antibody targeting VEGF-A indicated in metastatic breast cancer, combined with paclitaxel or capecitabine. It is administered by intravenous (IV) injections every 2 to 3 weeks. The side effects that may be observed are arterial hypertension (incidence of grade III / IV hypertension ≃ 4%), proteinuria (presence of protein in the urine), an increased incidence of arterial thromboembolic events ( ≃ 3.8%) or even heart failure.
- Trastuzumab is an HER2 blocking monoclonal antibody. It has many indications in breast cancer: non-metastatic, after the use of or associated with cytotoxics (taxanes, alkylating agents, such as carboplatin), or metastatic associated with taxane and hormone therapy. It can be injected intravenously or subcutaneously. The side effects that may be observed are dyspnea or cardiomyopathy (a cardiac ultrasound before the start of treatment is necessary) and therefore cannot be combined with anthracyclines.
- Lapatinib (Tyverb®) is an EGFR receptor tyrosine kinase (TKI) inhibitor. It is indicated in breast cancer with overexpression of HER2 receptors in combination, depending on the situations and the treatments previously received, with capecitabine, trastuzumab or an aromatase inhibitor. It is used orally. The side effects which may be observed are diarrhea, hand-foot syndrome, skin rash, cardiotoxicity and pulmonary disorders (interstitial damage and pneumonia).
Other treatments used are:
- Pertuzumab (Perjeta®), indicated in metastatic or locally recurrent HER2 + breast cancer in combination with trastuzumab and docetaxel in adults who have not previously received anti-HER2 treatment or chemotherapy for their metastatic disease.
- T-DM1 (trastuzumab-emtansine) (Kadcyla®), an ADC (antibody-drug conjugate or Antibody-Drug Conjugate), indicated in the monotherapy treatment of HER2 + breast cancers, locally advanced or metastatic, unresectable, for patients who have been treated with trastuzumab and taxane, separately or in combination.
- Everolimus, a selective inhibitor of mTOR (serine-threonine kinase whose activity is known to be deregulated in many cases of human cancers), indicated in the treatment of advanced breast cancer with positive hormone receptors, HER2 negative, in combination with exemestane, in postmenopausal women.
- CDK inhibitors (cyclin-dependent protein kinases, responsible for triggering and coordinating the different phases of the cell division cycle). Among them are palbociclib (Ibrance®), ribociclib (Kisqali®) and abemaciclib (Verzenois®). All three are CDK 4/6 inhibitors, used in RH + HER2- metastatic breast cancer, in combination with letrozole or fulvestrant.
Living with breast cancer
Medical care
Close medical follow-up for several years is necessary for women who have received treatment for breast cancer, in order to detect the onset of late side effects and to quickly screen for any relapse. This follow-up is carried out by a multidisciplinary team composed, among others, of a general practitioner, a gynecologist, an oncologist, a radiologist, a surgeon, a nutritionist or a psychologist.
The classic scheme includes follow-up visits every 3 to 4 months, then every 6 months for 5 years, then once a year. However, this visiting schedule must be discussed and adapted to each patient, in particular according to the severity of the cancer and the risk of relapse. In fact, for women who have suffered from metastatic breast cancer, follow-up is more intense, every 2 to 3 months or every 2 to 3 courses of chemotherapy, if it is administered over a long period of time.
The first follow-up mammogram should be performed 1 year after the initial mammogram and at least 6 months after the end of radiotherapy.
Finally, breast reconstruction requires a follow-up visit with the surgeon who performed it, every year or every 2 years.
Healthy lifestyle
In order to be better prepared to fight against the progression of cancer and the side effects of treatments, it is important to have a balanced diet, to eat food with anti-inflammatory properties and rich in antioxidants: fruit and vegetables, olive oil, fatty fish, nuts ... If cancer is treated with hormone therapy, vitamin D and calcium supplements may be helpful in preventing osteoporosis.
It is also essential to practice a regular physical activity (cycling, swimming, walking, gardening, cleaning ...). It is proven that a well-adapted, regular and moderate physical activity can reduce fatigue caused by treatments.
Psychological support
Communicating, sharing your doubts and fears about the disease (new side effects, questions about a new treatment) with your doctor, but also with those around you, is important. It may also be beneficial to turn to support groups, associations of patients with the same disease. You can also join online patient communities, like Carenity Breast Cancer Forum. If necessary, psychological support (psychologist, psychiatrist, etc.) can be considered.
Getting back to work
A sick leave, the duration of which depends on the type of treatment and on post-operative complications, may be prescribed by your GP. In addition, reasonable workplace or job role adjustments can be considered, such as working reduced hours, or some workstation adjustments, for example. This can be discussed with your medical team, occupational health professional or your HR department.
In conclusion, breast cancer is the most common cancer in women. However, detected early, it can be cured in 9 out of 10 cases. It is therefore important to perform self-examination and regular screening. There are many treatments, used alone or in combination. Adapted to each patient (according to their clinical, biological and genetic characteristics), they make it possible, despite the side effects, to improve the quality of life and the overall survival rate of patients.
Sources :
Le cancer du sein : points clés, Institut national du cancer
Comprendre le cancer du sein, Ameli
Qu’est-ce que le cancer du sein?, Société canadienne du cancer
Cancer du sein, Santé publique france
Qu’est-ce que le cancer du sein ?, Vidal
Cancer du sein, la ligue contre le cancer
Cancers du sein: les traitements, fondation ARC
Comprendre le cancer du sein, fondation pour la recherche médicale
Published Mar 18, 2018 • Updated Oct 30, 2021
Breast cancer community
Breakdown of 1 357 members on Carenity
Average age of our patients
Fact sheets
See more